A complete introduction of Hastelloy B,C,G alloy for chemical industrial
Ultra-low carbon Ni-Cr-Mo alloy is known as Hastelloy alloy. Hastelloy is a trademark of Haynes International company, consist of the letter “HA” in Haynes, “STELL” in STEILITE and “OY” in ALLOYS. As an advanced nickel-based alloy, Hastelloy offers excellent corrosion resistance to a variety of harsh corrosion environments such as wet oxygen, sulfurous acid, strong oxidizing salt medium, etc.
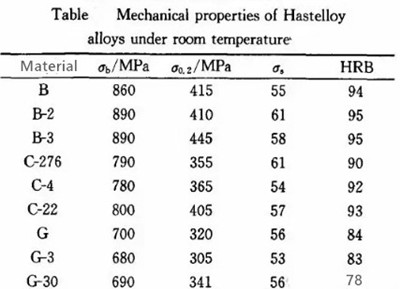
Hastelloy alloy has many series like Hastelloy A, B, C, D, F, G, N, W and X. The main companies producing Hastelloy alloy series are American Haynes International Inc (Hastelloy alloy research and development company), Special Metal (superalloy group) and ThyssenKrupp company of Germany. The alloys used in chemical plants are mainly Hastelloy B, C, and G, which mechanical properties are shown in the following table.
1)Hastelloy B
Series B includes Hastelloy B, Hastelloy 132, Hastelloy B3, etc. Hastelloy B alloy has the best corrosion resistance to hydrochloric acid,reducing its content of carbon to 0.02% and Si to 0.1%. that becomes Hastelloy b-2 alloy. Hastelloy B-2 can be used in hydrochloric acid medium of any concentration at boiling temperature and improves the resistance to intercrystalline corrosion in sensitization and post-welding.
In the 1990s, the invention of Hastelloy b-3 completely changed the defect of b-2 which was that it was easy to precipitate ni-mo precipitate at an intermediate temperature and improved the processing performance. However, Hastelloy B and Hastelloy B-2 alloys contain very low Cr and cannot be used in oxidizing environments. The corrosion resistance of this kind of Harley alloy under typical environment is shown in the table below.
| Medium | Average corrosion resistance rate of Hastelloy B alloys (mm/a) | |||
| B-3 | B-2 | 316L | Monel400 | |
| 50% Acetic acid | 0.005 | 0.01 | 0.005 | / |
| 40% Formic acid | 0.013 | 0.018 | 1.041 | 0.053 |
| 55% Phosphoric acid | 0.076 | 0.152 | 0.457 | 0.114 |
| 50% Sulfuric acid | 0.043 | 0.030 | >500 | 4.699 |
2)Hastelloy C
Ni-Cr-Mo alloy Hastelloy C is a universal corrosion-resistant material suitable for all kinds of environments. The content of the C-series alloy is high. Element Cr and Mo play the role of oxidation-resistant medium and reducing medium corrosion respectively to resist local corrosion (pitting corrosion and crevice corrosion).
Hastelloy C alloy group consists of C, C-276, C-4, C-22 and C-2000. The first alloy Hastelloy C was produced in the 1930s, then C-4 in the 1970s, C-22 In the 1980s; alloy 59, 686, C-2000 and so on In the 1990s. Intercrystal corrosion, local corrosion, stress corrosion and passivation of Ni-Cr-Mo alloys were studied by electrochemical methods. S Ghosh study showed that C-276 alloy had better corrosion resistance than UNS N08367 and UNS N08028 in NaCl and H3PO4 solutions at 95℃. The corrosion resistance of class C alloys under typical conditions is shown in the table.
| Medium | Corrosion resistance rate of Hastelloy C alloys in different boiling medium(mm/a) | ||||
| C-276 | C-22 | 686 | C-2000 | C-59 | |
| ASTM 28A | 6.10 | 0.91 | 2.62 | 0.69 | 0.61 |
| ASTM 28B | 1.40 | 0.18 | 0.25 | 0.10 | 0.10 |
| 10%HNO3 | 0.48 | 0.05 | / | / | 0.05 |
| 15%HNO3 | 19.05 | 1.32 | 5.87 | / | 1.02 |
| 50%H2SO4 | 6.10 | 7.82 | / | / | 4.47 |
| 5%HCL | 0.28 | 0.36 | 0.13 | 0.038 | 0.08 |
| 10H2SO4+1%HCL | 2.22 | 8.99 | / | / | 1.78 |
(3)Hastelloy G
Ni-Cr-Cu-Mo alloy Hastelloy G, which is more resistant to sulfuric acid and phosphoric acid than Hastelloy C alloy. The alloy with high cr content has good corrosion resistance in sulfuric acid, phosphoric acid, sulfuric acid, phosphoric acid and wet-process phosphoric acid-containing F- and Cl-. Elements adding such as Si and B are able to withstand corrosion by concentrated sulfuric acid in HAS G alloy, which is characterized by high hardness and good abrasion resistance.
G series includes G, G-3, G-30, G-50 and etc. The birth of Hastelloy G-3, G-30 alloy solved the application of alloy in strong oxidation or mixed acid environment. The new Hastelloy G-35 is essentially a Ni-Cr-Mo alloy with a composition close to that of a C-series alloy. Because it is designed for strong oxidation and mixed acid environments and it is still included in the G series.


